 So after the neutron breaks down, and before emission, there should now be: 11 neutrons, 7 protons, 7 electrons and 1 anti-neutrino. When it decays 1 neutron is converted into: 1 proton, 1 electron and 1 anti-neutrino. See also What is the formula for time travel? How many electrons does carbon-14 have after beta decay?Ĭarbon-14 has 12 neutrons, 6 protons and 6 electrons. 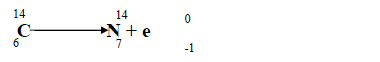
One type (positive beta decay) releases a positively charged beta particle called a positron, and a neutrino the other type (negative beta decay) releases a negatively charged beta particle called an electron, and an antineutrino. What produces carbon-14?Ĭarbon-14 is produced in the stratosphere by nuclear reactions of atmospheric nitrogen with thermal neutrons produced naturally by cosmic rays (with the highest production rate 10 to 13 miles above Earth’s poles), as well as by atmospheric nuclear weapons testing in the 1950s and ’60s. The carbon-14 nucleus has six protons and eight neutrons, for an atomic mass of 14. What type of radioisotope is carbon-14?Ĭarbon-14, the longest-lived radioactive isotope of carbon, whose decay allows the accurate dating of archaeological artifacts. The amount of Carbon 14 contained in a preserved plant is modeled by the equation f(t) = 10e^. What is the formula for carbon-14 decay?Ĭarbon 14 is a common form of carbon which decays over time. The beta radiation barely penetrates the outer protective dead layer of the skin of the body. Is carbon-14 alpha or beta?Ĭarbon-14 is a low energy beta emitter and even large amounts of this isotope pose little external dose hazard to persons exposed. In beta minus decay, a neutron decays into a proton, an electron, and an antineutrino: n Æ p + e – +. See also Why is Proxima Centauri b habitable? What is meant by carbon-14 is a beta emitter?Ĭarbon 14 is unstable and undergoes radioactive decay by emitting a beta particle (an electron) and an antineutrino (zero mass, zero charge) and becoming nitrogen (seven protons): What is the beta decay of isotopes?īeta decay occurs when, in a nucleus with too many protons or too many neutrons, one of the protons or neutrons is transformed into the other. The carbon-14 in presence in the object undergoes beta decay, transforming into a stable isotope of nitrogen. 
What is the product made when C 14 undergoes beta decay?īeta Decay of Carbon-14: Carbon-14 is one of the known radioactive isotopes of carbon, and its radioactive properties are utilized for estimating the age of an object that contains organic materials. What isotope is produced by the beta decay of carbon-14 _?Ĭarbon-14 undergoes beta decay to the stable nitrogen-14 isotope. This increases the number of protons in the atom by one, creating a nitrogen atom rather than a carbon atom. What is the formula for the decay rate of carbon-14?īeta Decay During this process, an atom of 14C decays into an atom of 14N, during which one of the neutrons in the carbon atom becomes a proton.What is the decay constant of carbon-14?. 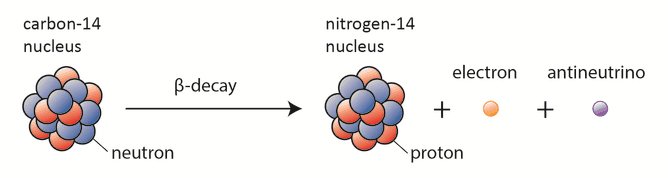
How many electrons does carbon-14 have after beta decay?.What type of radioisotope is carbon-14?.What is the formula for carbon-14 decay?.What is meant by carbon-14 is a beta emitter?.What is the product made when C 14 undergoes beta decay?.What isotope is produced by the beta decay of carbon-14 _?.What is the decay of carbon-14 by beta?.These nuclides lie below the band of stability. Positron emission is observed for nuclides in which the n:p ratio is low. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
